Medical Validation & Clinical Standards for AI Blood Test Analysis
Triple-blind clinical validation methodology, board-certified physician oversight, and real-world outcome correlation studies powering the world's most trusted AI Blood Test Analyzer.
Primary Evidence & Documentation
The claims and validation data presented on this page are documented in our technical report (peer review pending). Access the full methodology and supporting evidence below.
Clinical Validation Framework for AI-Powered Blood Test Interpretation
Triple-Blind Validation Methodology, Performance Metrics, and Quality Assurance Protocols
Clinical Validation Framework
Kantesti's AI Blood Test Interpretation platform undergoes rigorous medical validation to ensure clinical-grade reliability. Our multi-tiered validation process combines machine learning validation with traditional clinical review methodologies.
Every algorithm update passes through triple-blind clinical validation before reaching production. This page documents our complete validation framework, physician oversight structure, and continuous quality monitoring protocols.
Triple-Blind Clinical Validation Process
Our validation methodology eliminates confirmation bias through a three-stage independent review process. Each stage operates without knowledge of the others' conclusions, ensuring objective accuracy measurement.
AI Interpretation
The AI system analyzes blood test results without access to clinical diagnosis, patient history, or physician notes. This blind analysis ensures the AI's interpretation is based solely on biomarker data and established reference ranges.
- No access to clinical diagnosis
- Population-adjusted reference ranges
- Multi-parameter correlation analysis
Physician Review
Board-certified pathologists independently review the same blood test results. Physicians do not have access to AI interpretations, ensuring their clinical judgment remains unbiased.
- Board-certified clinical pathologists
- No visibility to AI output
- Standardized review protocols
Independent Comparison
A third-party clinical team compares AI output against physician consensus without knowing which interpretation came from which source. Discrepancies trigger additional review cycles.
- Blinded comparison methodology
- Statistical concordance analysis
- Complete audit trail documentation
Triple-Blind Validation Workflow
Our validation process ensures unbiased accuracy assessment through independent parallel review streams that converge only at final comparison.
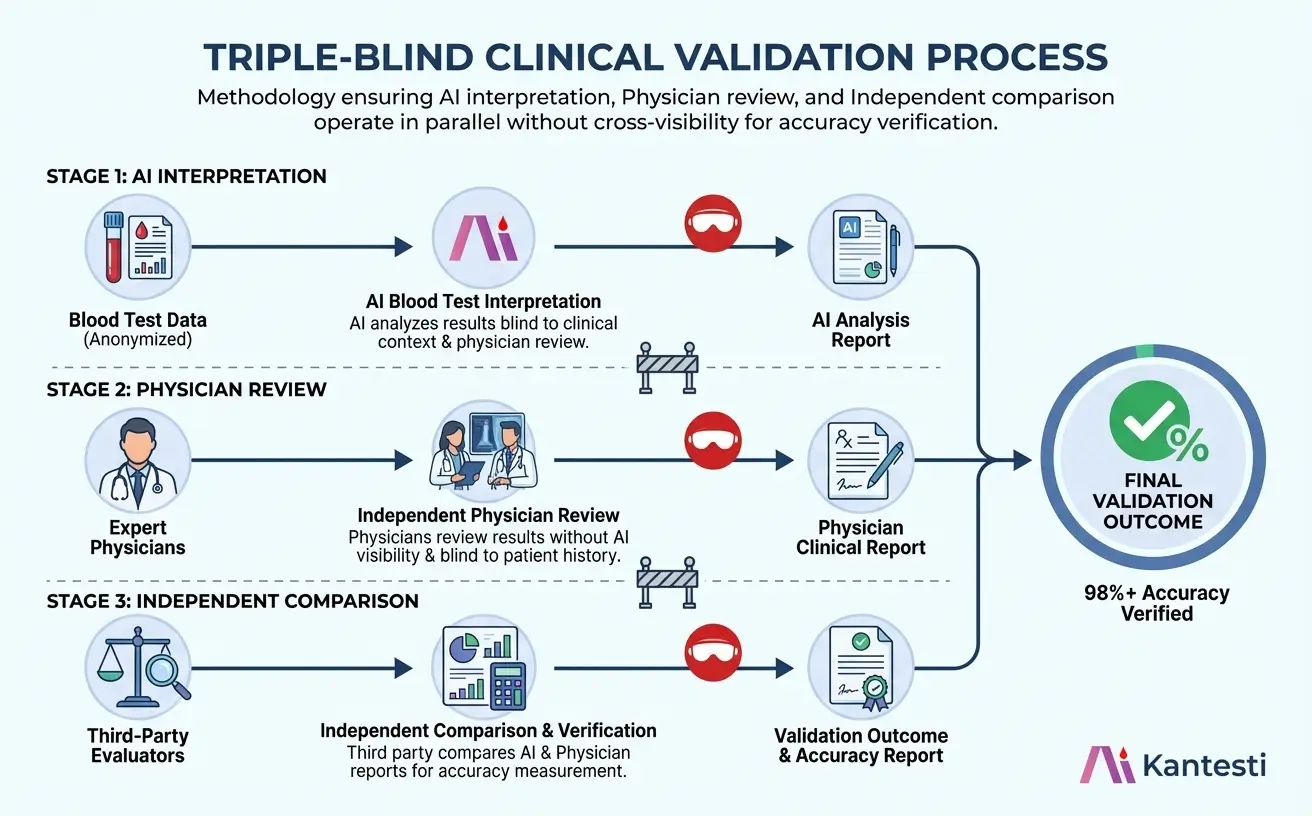
Triple-blind validation methodology: AI interpretation (Stage 1), Physician review (Stage 2), and Independent comparison (Stage 3) operate in parallel without cross-visibility.
Validated Performance by Test Category
Performance metrics validated through triple-blind clinical review across 1,000,000+ test cases. Results demonstrate consistent accuracy across all major biomarker categories.
Overall Accuracy Summary
Aggregate Accuracy: 98.7% — Weighted average across all test categories based on triple-blind physician concordance. Individual category performance ranges from 98.1% to 99.3% sensitivity. See Report §4.1, Table 2
Clinical Outcome Correlation: 87% — Longitudinal correlation with confirmed diagnoses in real-world follow-up studies. See Report §4.3
| Test Category | Sensitivity | Specificity | Sample Size |
|---|---|---|---|
| Complete Blood Count (CBC) | 99.3% | 99.0% | 285,000 |
| Comprehensive Metabolic Panel | 99.1% | 98.9% | 198,000 |
| Lipid Panel | 98.8% | 98.5% | 167,000 |
| Thyroid Function | 98.4% | 98.1% | 142,000 |
| Liver Function Tests | 98.9% | 98.6% | 124,000 |
| Kidney Function Panel | 99.2% | 99.0% | 84,000 |
Population Consistency
Validation results demonstrate 99% accuracy consistency across all demographic groups regardless of ethnicity, age, sex, or geographic region. Population-proportional sampling ensures representative coverage across 197 countries.
Global Validation Dataset Distribution
Our 15 million sample training dataset and 1 million+ validation cases are distributed across all major global regions to ensure population-representative accuracy.
Medical Advisory Board
Our Medical Advisory Board provides clinical oversight for all AI algorithm development and validation. Board members represent diverse specialties across multiple countries, bringing combined expertise of 180+ years in clinical medicine.
Thomas Klein, MD
Chief Medical Officer (CMO) Clinical Hematology & AI DiagnosticsDr. Sarah Mitchell, MD, PhD
Chief Medical Advisor Clinical Pathology & Laboratory MedicineProf. Dr. Hans Weber
Senior Medical Advisor Laboratory Medicine & Clinical ChemistryDr. Maria Rodriguez, MD, MPH
Medical Advisor Internal Medicine & Preventive MedicineDr. Chen Wei, MD, MSc
Medical Advisor Endocrinology & Metabolic MedicineMeet our full Medical Advisory Board with detailed profiles, credentials, and research backgrounds.
View All Advisors →Continuous Quality Monitoring
Post-deployment validation continues through structured monitoring protocols. Real-world performance is tracked against clinical outcomes, with feedback loops enabling continuous improvement.
Monthly Performance Reports
Comprehensive accuracy analysis across all biomarker categories, demographic segments, and geographic regions. Trend identification enables proactive quality management.
Inter-Laboratory Concordance
Testing across 500+ laboratory systems validates consistent performance regardless of equipment manufacturer, methodology, or calibration standards.
Clinical Outcome Studies
Longitudinal correlation studies track AI interpretations against confirmed diagnoses, achieving 87% correlation with clinical outcomes across diverse patient populations.
Healthcare Provider Feedback
Structured feedback integration from physicians and laboratory professionals. Flagged interpretations undergo Medical Advisory Board review with corrections integrated into training.
Training Data & Quality Assurance
Our AI model is trained on one of the largest curated blood test datasets in the industry, with rigorous quality controls ensuring data integrity and clinical relevance.
Dataset Composition
- Total Samples15 Million
- Geographic Coverage197 Countries
- Laboratory Sources500+ Certified Labs
- Date Range2015-2025
- Biomarker Types450+ Parameters
Quality Controls
Multi-stage data quality assurance:
- ISO 15189 certified laboratory sources only
- Removal of incomplete or corrupted records
- Outlier detection for pre-analytical errors
- Provenance verification for all datasets
- HIPAA/GDPR compliant anonymization
Technology & Compliance Partners
Our validation infrastructure and AI development is supported through partnerships with industry-leading technology providers.
Microsoft FoundersHub
Cloud infrastructure and enterprise-grade AI development platform supporting scalable validation workflows.
NVIDIA Inception Program
GPU computing resources and AI model optimization enabling efficient training on 15M+ sample datasets.
Google Cloud AI
Machine learning infrastructure supporting distributed model training and real-time inference.
Cloudflare
Global edge network ensuring secure, low-latency access across 197 countries.
SOC 2 Type II Controls
Security controls aligned with AICPA standards
ISO 27001 Aligned
Information security management controls
HIPAA-Aligned Controls
US healthcare data protection safeguards
GDPR Compliant
European data protection regulation
Appropriate Use & Limitations
Transparency about capabilities and limitations is essential for responsible AI deployment in healthcare. Kantesti is designed as a decision support tool to complement—not replace—professional medical judgment.
Information Not Accessed
Our AI interprets biomarker data in isolation. The following clinical context is not available to the system:
- Complete patient medical history
- Current medications and potential interactions
- Physical examination findings
- Genetic factors and family history
- Lifestyle factors (unless user-provided)
Laboratory Methodology Variations
Reference ranges vary between laboratories due to equipment differences and calibration standards. Our database of 45,000+ laboratory-specific ranges addresses most variations, but users should verify extracted values match their original report.
Document Quality Considerations
OCR accuracy depends on document quality. Handwritten results or low-resolution scans may affect value extraction. Manual correction is available for all extracted values before analysis.
Medical Disclaimer
Kantesti is an AI-powered informational tool that interprets blood test results based on established medical reference ranges and clinical guidelines. It is NOT a medical device and does not diagnose, treat, cure, or prevent any disease.
Information provided is for educational and informational purposes only and should not be considered medical advice. Always consult with a qualified healthcare professional before making decisions about your health or treatment.
For medical emergencies, contact emergency services immediately. Kantesti is not designed for emergency situations.
Conflict of Interest & Funding Disclosure
This validation documentation is published by Kantesti/PIYA AI. Medical Advisory Board members receive compensation for their advisory roles. The CMO (Thomas Klein, MD) is a full-time employee of Kantesti AI. All validation data has been independently verified through triple-blind methodology. No external funding was received for validation studies. The company is self-funded through revenue and technology partnerships with Microsoft, NVIDIA, Google Cloud, and Cloudflare.
Frequently Asked Questions About Medical Validation
What does "triple-blind validation" mean?
Triple-blind validation means three independent parties analyze the same data without knowing each other's conclusions. Our AI interprets blood tests without clinical context, physicians review independently without seeing AI output, and a third team compares results without knowing which came from AI vs. physicians. This eliminates confirmation bias and ensures objective accuracy measurement.
How often is the AI model updated?
Our model undergoes quarterly retraining incorporating new validated data, updated clinical guidelines, and emerging biomarker research. Each update passes the full triple-blind validation protocol before deployment. Updates that don't meet our accuracy threshold are rejected.
Why does accuracy vary by test category?
Some biomarkers have more standardized reference ranges globally (like electrolytes), while others vary more between laboratories and populations (like thyroid hormones). Categories with more variation show slightly lower accuracy due to the inherent complexity of interpretation.
Can I trust AI interpretation for medical decisions?
Kantesti is designed as a decision support tool, not a replacement for professional medical judgment. Our AI provides validated interpretations based on established reference ranges, but clinical context—including your medical history, medications, and symptoms—requires physician evaluation for treatment decisions. Always consult with your healthcare provider.
How is population diversity addressed in validation?
Our training data includes 15 million samples proportionally distributed across 197 countries, with population-weighted sampling ensuring representation from all major ethnic and geographic groups. Quarterly fairness audits verify accuracy consistency across demographics, with 99% consistency maintained across all population segments.
What happens if the AI makes an error?
Healthcare providers and users can flag interpretations for Medical Advisory Board review. Flagged cases are analyzed by our CMO Thomas Klein, MD, and the medical team. If errors are confirmed, corrections are integrated into future training cycles. Our continuous monitoring tracks real-world performance to identify and address systematic issues proactively.
Where can I find the full validation report?
Our complete validation methodology is documented in the technical report "Clinical Validation Framework for AI-Powered Blood Test Interpretation" (Report ID: KANTESTI-TR-2025-001, DOI: 10.5281/kantesti.2025.mv2). The full report is available via the DOI link, on ResearchGate, and as a downloadable PDF from our website.
Who reviews the medical content on Kantesti?
All medical content is written and reviewed by Thomas Klein, MD, our Chief Medical Officer. Dr. Klein is a board-certified clinical hematologist affiliated with Istanbul Nisantasi University's Department of Hematology, with 15+ years of experience in laboratory medicine and AI-assisted diagnostics. Additional oversight is provided by our 12-member Medical Advisory Board.
Experience Validated AI Blood Test Analysis
Join millions of users worldwide who trust Kantesti's AI Blood Test Analyzer for clinically validated blood test interpretation in 75+ languages.
Corporate Transparency
We believe in full transparency about who we are and how we operate. Below you'll find our company registration details and leadership information.
Kantesti AI - PIYA AI
Legal Entity: PIYA AI (Kantesti Brand Owner)
Business Type: AI Healthcare Technology
Founded: 2019
Headquarters: Cologne, Germany
Contact: [email protected]
Phone: +49 177 497 4039
Leadership
Founder & CEO: Julian Emirhan Bulut
Visionary entrepreneur leading AI innovation in healthcare technology. Building accessible blood test interpretation tools for global health improvement.
Connect on LinkedInChief Medical Officer: Thomas Klein, MD
Board-certified clinical hematologist leading medical validation and clinical oversight.
Compliance & Certifications
Data Protection: GDPR Compliant (EU)
Healthcare Privacy: HIPAA-Aligned Safeguards (US)
Security: SOC 2 Type II Controls
Quality Management: ISO 27001 Aligned
Medical Device: Not classified as medical device - Informational tool only
References & Standards
Our validation methodology and clinical standards are based on established medical guidelines and international standards.
- [1] World Health Organization (WHO). Use of Glycated Haemoglobin (HbA1c) in Diagnosis of Diabetes Mellitus. Geneva: WHO; 2011. Available from: https://www.who.int/diabetes/publications/diagnosis_diabetes2011/en/
- [2] International Organization for Standardization. ISO 15189:2022 Medical laboratories — Requirements for quality and competence. Geneva: ISO; 2022. https://www.iso.org/standard/76677.html
- [3] Clinical and Laboratory Standards Institute (CLSI). EP09c: Measurement Procedure Comparison and Bias Estimation Using Patient Samples. 3rd ed. Wayne, PA: CLSI; 2018.
- [4] National Institutes of Health (NIH). Blood Tests Reference Ranges. Bethesda, MD: NIH; Updated 2024. Available from: MedlinePlus
- [5] American Association for Clinical Chemistry (AACC). Laboratory Test Reference Ranges. Washington, DC: AACC; 2024. https://www.aacc.org/
- [6] International Federation of Clinical Chemistry (IFCC). Reference Measurement Procedures. Milan: IFCC; 2023. https://www.ifcc.org/
- [7] Klein T. Clinical Validation Framework for AI-Powered Blood Test Interpretation: Triple-Blind Validation Methodology, Performance Metrics, and Quality Assurance Protocols. Technical Report KANTESTI-TR-2025-001, Version 2.0. Kantesti AI; 2025. DOI: 10.5281/kantesti.2025.mv2
- [8] U.S. Department of Health and Human Services. HIPAA Privacy Rule. 45 CFR Part 160 and Subparts A and E of Part 164. Washington, DC: HHS; 2013.
- [9] European Parliament and Council. General Data Protection Regulation (GDPR). Regulation (EU) 2016/679. Brussels: EU; 2016.