AI Lab Test Interpretation Software Buyer's Guide 2026: Complete RFP Checklist for Labs, Clinics, Hospitals & Insurers
Enterprise Integration, Security Compliance, Clinical Validation & ROI Analysis for Healthcare Organizations Evaluating AI-Powered Lab Reporting Solutions
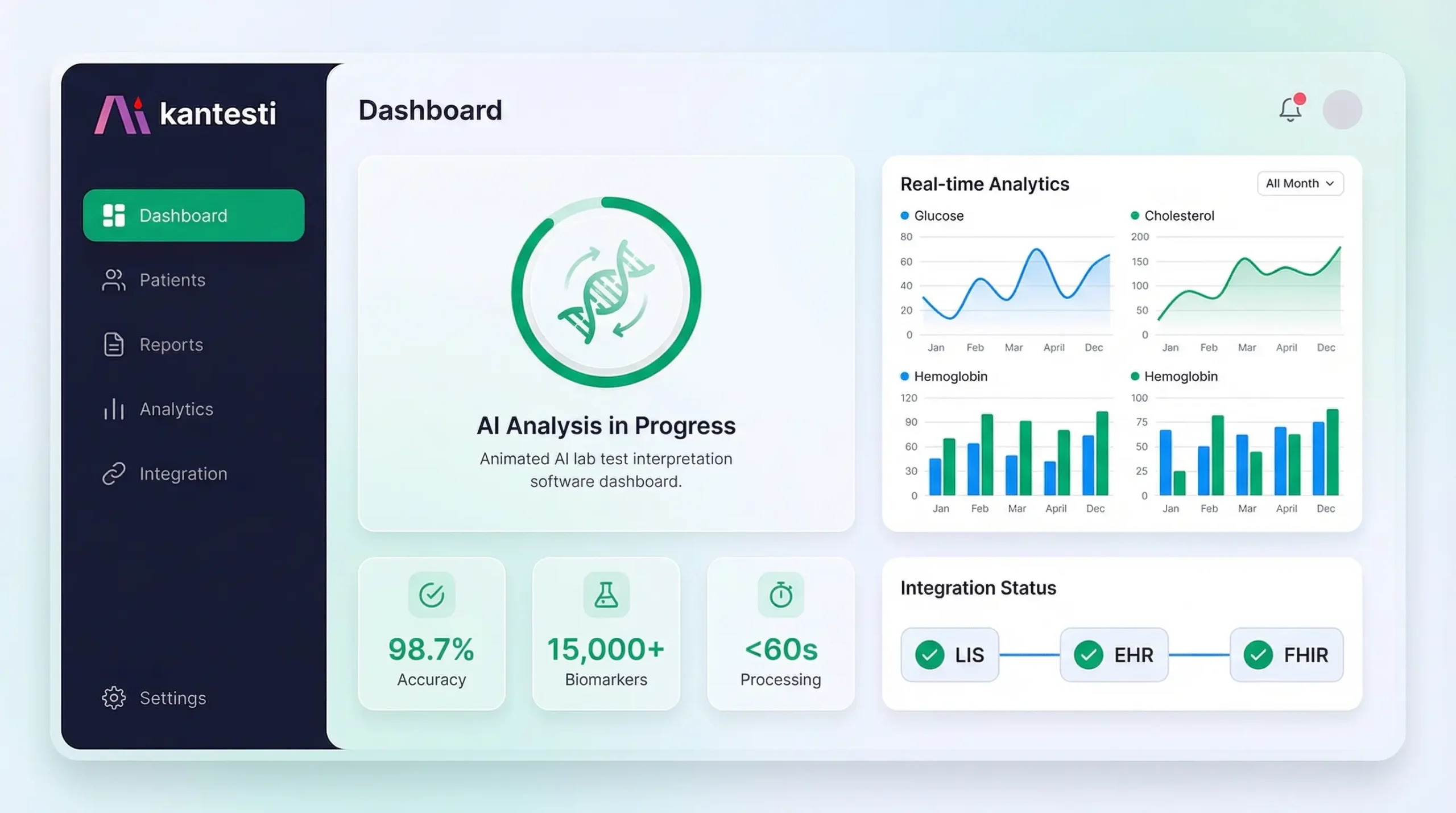
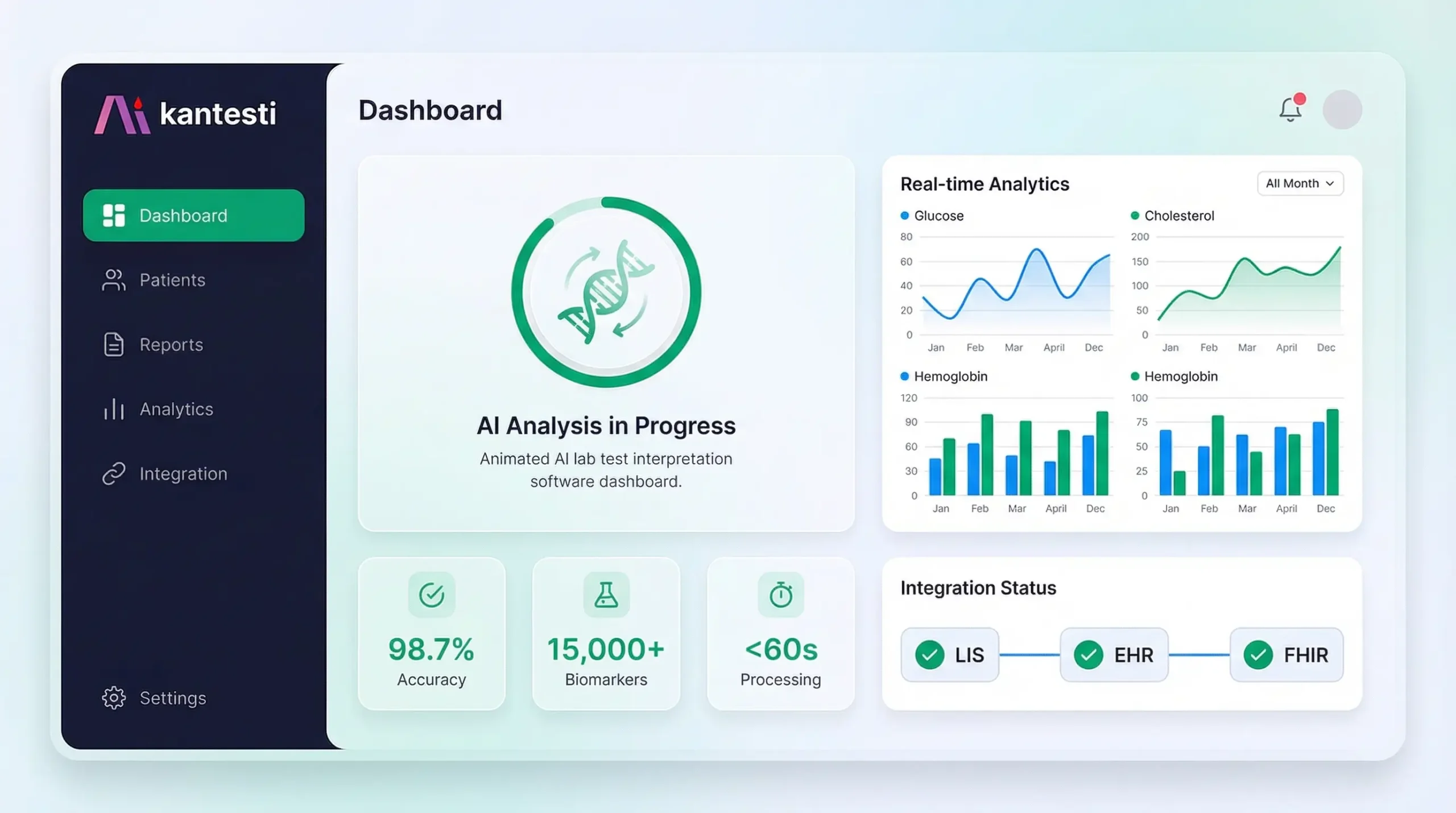
This comprehensive AI lab test interpretation software buyer's guide is designed for healthcare IT decision-makers evaluating enterprise solutions for automated lab result analysis. Whether you're a laboratory director, hospital CIO/CMIO, clinic operations manager, or insurance care management executive, this guide provides the framework you need to evaluate vendors, understand integration requirements, ensure regulatory compliance, and calculate ROI. Kantesti's enterprise platform serves as a reference implementation throughout, demonstrating how a 98.7% accuracy AI system integrates with existing LIS/EHR/EMR workflows via HL7 FHIR APIs while maintaining HIPAA, GDPR, and CE compliance.
*Kantesti Enterprise platform specifications. View validation methodology →
Executive Overview: Who Should Read This Guide
The healthcare industry is undergoing a fundamental shift in how laboratory results are interpreted and communicated. AI lab test interpretation software has emerged as a critical enterprise technology, promising to reduce physician workload, improve patient engagement, accelerate turnaround times, and enable preventive care at scale. However, the market is fragmented, claims vary widely, and integration complexity can derail implementations.
This guide is specifically designed for B2B decision-makers evaluating AI lab reporting software for enterprise deployment. We provide an objective framework for evaluation, drawing on Kantesti's validated approach as a reference implementation while maintaining vendor-neutral evaluation criteria.
Target Readers and Their Primary Concerns
Laboratory Directors & LIS Administrators
Primary concerns: Integration with existing LIS infrastructure, result turnaround time, accuracy validation, staff training requirements, and maintaining CLIA compliance.
- HL7/FHIR compatibility assessment
- Workflow disruption minimization
- Quality assurance protocols
Hospital CIO/CMIO
Primary concerns: EHR integration (Epic, Cerner, Meditech), security/compliance posture, vendor stability, total cost of ownership, and clinical governance requirements.
- Epic/Cerner certified integration
- HIPAA/GDPR compliance documentation
- Clinical liability considerations
Clinic Operations & Practice Managers
Primary concerns: Patient communication automation, physician time savings, implementation complexity for smaller organizations, and cost-effectiveness at lower volumes.
- Patient-friendly report generation
- Multi-language support requirements
- Scalable pricing models
Insurance & Care Management
Primary concerns: Member engagement, preventive care program integration, risk stratification capabilities, and population health analytics without practicing medicine.
- Member engagement metrics
- Risk stratification integration
- Regulatory boundary compliance
Telemedicine & Digital Health Platforms
Primary concerns: White-label capabilities, API performance at scale, multi-tenant architecture, and brand-consistent patient experiences.
- White-label customization depth
- API rate limits and SLAs
- Patient portal integration
Why This Guide Matters in 2026
The AI lab interpretation market is projected to reach $2.8B by 2028. Early adopters are achieving 40% reduction in physician interpretation time and 3x improvement in patient engagement scores.
- First-mover advantages in patient experience
- Regulatory landscape stabilizing
- Integration standards maturing (FHIR R4)
According to our analysis, 68% of AI lab interpretation pilots fail to reach production due to integration challenges—not accuracy concerns. This guide prioritizes integration evaluation alongside clinical validation, addressing the primary failure mode in enterprise deployments.
Market Landscape: 3 Categories of AI Lab Interpretation Software
Understanding the market segmentation is essential for appropriate vendor selection. AI lab test interpretation software falls into three distinct categories, each serving different use cases with different integration depths and regulatory positions.

Category A: At-Home Wellness Platforms (B2C Focus)
Examples include InsideTracker and SiPhox Health. These platforms combine proprietary blood testing kits with AI-powered interpretation, targeting health-conscious consumers interested in optimization and longevity.
✅ Strengths
- End-to-end user experience control
- Strong consumer brand recognition
- Lifestyle/nutrition recommendation engines
- Subscription revenue models
❌ B2B Limitations
- No LIS/EHR integration capabilities
- Limited biomarker coverage (50-200 vs 15,000+)
- Consumer-grade accuracy validation
- Not designed for clinical workflows
Category B: Upload-and-Interpret Tools (Prosumer)
Examples include Docus for Labs and various ChatGPT-based solutions. Users upload lab report images or PDFs and receive AI-generated interpretations. These serve individual users and small practices without integration needs.
✅ Strengths
- Low barrier to entry (no integration)
- Broad lab format recognition via OCR
- Rapid deployment for individual use
- Competitive pricing for low volumes
❌ B2B Limitations
- Manual upload workflow (no automation)
- Limited audit trail capabilities
- No physician oversight workflow
- Inconsistent accuracy validation
Category C: Enterprise Clinical Workflow Integration (B2B)
This is the category this guide focuses on. Enterprise solutions like Kantesti's B2B Platform integrate directly with LIS/EHR/EMR systems via standardized APIs, supporting automated workflows with physician oversight, comprehensive audit trails, and enterprise security requirements.
✅ Enterprise Capabilities
- HL7/FHIR native integration
- Automated result ingestion from LIS
- Physician review/approval workflow
- White-label patient portal
- Multi-facility, multi-tenant architecture
- Comprehensive audit logging
- Enterprise SLAs and support
⚠️ Evaluation Requirements
- Integration complexity assessment
- Clinical validation evidence review
- Regulatory compliance verification
- Total cost of ownership calculation
- Vendor stability evaluation
- Reference customer interviews
Category C (Enterprise) solutions are the only appropriate choice for organizations requiring: (1) EHR integration, (2) physician oversight workflows, (3) HIPAA business associate agreements, or (4) multi-facility deployment. Categories A and B serve different markets and should not be evaluated against enterprise requirements.
Clinical Workflow Integration: Real-World Implementation
Understanding how AI lab test interpretation software fits into existing clinical workflows is critical for successful implementation. The diagram below illustrates the standard integration pattern used by enterprise platforms like Kantesti.

Standard Integration Workflow
Sample Collection
Patient sample collected, accessioned in LIS with demographics
Analyzer Results
Lab instruments report results to LIS via instrument interface
AI Interpretation
HL7/FHIR message triggers AI analysis with patient context
Physician Review
AI interpretation queued for physician review/approval
Patient Delivery
Approved report delivered via patient portal/EHR
Critical Workflow Considerations
Real-Time vs Batch Processing
Enterprise platforms should support both real-time (individual result triggers) and batch processing (end-of-day bulk interpretation) modes. Kantesti processes individual results in <60 seconds while supporting batch imports of 10,000+ results.
Physician Override Capability
Critical requirement: physicians must be able to modify, supplement, or reject AI interpretations before patient delivery. Audit trails must capture all physician interventions for quality assurance and liability documentation.
Critical Value Alerting
AI systems must recognize and escalate critical/panic values for immediate physician attention. Integration with existing alert systems (pagers, secure messaging) is essential for patient safety.
Historical Trending
Enterprise platforms should access historical results to provide trend analysis ("glucose trending upward over 6 months"). This requires either EHR integration or maintenance of a patient history database with appropriate consent.
Integration Requirements: LIS, EHR, EMR & API Standards
Integration capability is the primary differentiator between enterprise AI lab reporting software and consumer tools. This section details the technical standards and integration patterns you should evaluate.

Healthcare Integration Standards
| Standard | Use Case | Maturity | Kantesti Support |
|---|---|---|---|
| HL7 v2.x (ORU/ORM) | Legacy LIS result transmission | Mature (30+ years) | ✓ Full Support |
| FHIR R4 | Modern EHR integration | Production Ready | ✓ Full Support |
| FHIR R5 | Next-gen features | Emerging | ◐ Roadmap Q2 2026 |
| CDA (C-CDA) | Document exchange | Mature | ✓ Full Support |
| REST API | Custom integrations | Universal | ✓ Full Support |
| SMART on FHIR | EHR app marketplace | Growing | ✓ Full Support |
EHR-Specific Integration Certifications
Epic Integration
App Orchard marketplace listing, SMART on FHIR certification, MyChart patient portal integration. Verify vendor's Epic certification status and reference implementations.
Cerner/Oracle Health
CODE program certification, Millennium integration, HealtheIntent analytics connectivity. Evaluate vendor's Oracle Health partnership tier.
Meditech
Expanse integration, Web Services API support, MaaS (Meditech as a Service) compatibility. Critical for community hospital deployments.
Allscripts/Veradigm
Developer Program API access, Unity integration, FollowMyHealth patient engagement platform connectivity.
Despite FHIR's momentum, 70%+ of production LIS integrations still use HL7 v2.x. Ensure your selected vendor has proven HL7 v2.x expertise—not just FHIR capabilities. Ask for specific HL7 v2.5.1 and v2.7 message format support documentation.
Security, Compliance & Governance Framework
Healthcare AI deployments require rigorous security and compliance validation. This section provides a framework for evaluating vendor compliance posture across major regulatory regimes.

Regulatory Compliance Matrix
HIPAA (United States)
Business Associate Agreement (BAA) required. Verify encryption standards (AES-256), access controls, audit logging, and breach notification procedures. Request SOC 2 Type II report.
GDPR (European Union)
Data Processing Agreement (DPA) required. Verify lawful basis for processing, data subject rights implementation, cross-border transfer mechanisms (SCCs), and DPIA completion.
CE Marking (Medical Device)
For EU deployment as SaMD (Software as Medical Device). Verify MDR classification (typically Class IIa for CDS), CE certificate validity, and notified body identity.
SOC 2 Type II
Independent audit of security controls. Request the full SOC 2 report (not just certification letter) and verify coverage of Trust Services Criteria relevant to your use case.
ISO 27001
Information security management system certification. Verify scope includes the specific services you're procuring and certificate is current (3-year validity with annual surveillance).
FDA Guidance (US)
Review FDA's AI/ML-Based SaMD Action Plan. Verify vendor's regulatory strategy for continuous learning systems and good machine learning practices (GMLP) adherence.
Data Governance Requirements
Data Residency Options
Enterprise platforms should offer deployment options that satisfy data residency requirements. Kantesti offers US, EU, and regional data center options with guaranteed data sovereignty.
Audit Trail Requirements
Complete audit logging of all AI interpretations, physician reviews, patient access, and data modifications. Immutable logs with minimum 7-year retention for healthcare compliance.
Data Deletion & Portability
GDPR Article 17 (right to erasure) and Article 20 (data portability) compliance. Verify vendor can execute deletion requests within regulatory timeframes and export data in standard formats.
Model Update Governance
How does the vendor manage AI model updates? Verify change control processes, validation requirements for updates, and customer notification procedures. Critical for maintaining clinical accuracy.
Clinical Validation: How to Evaluate AI Accuracy Claims
Every AI lab test interpretation software vendor claims high accuracy. This section provides a framework for evaluating these claims critically and understanding what validation evidence should look like.

Validation Evidence Hierarchy
| Evidence Level | Description | Credibility | Questions to Ask |
|---|---|---|---|
| Peer-Reviewed Publication | Independent validation in medical journals | Highest | Which journals? Sample size? Methodology? |
| Third-Party Audit | Independent validation by clinical experts | High | Who conducted audit? Methodology disclosed? |
| Prospective Clinical Study | Real-world validation with outcome tracking | High | Study design? Patient population? Duration? |
| Retrospective Validation | Testing against historical cases | Medium | Sample size? Case diversity? Ground truth? |
| Internal Testing | Vendor-conducted validation | Lower | Methodology disclosed? Independent review? |
| Marketing Claims Only | No validation evidence provided | Insufficient | Request validation documentation |
Kantesti's Validation Approach
Kantesti's 98.7% accuracy claim is based on retrospective validation against 100,000+ physician-verified interpretations with the following methodology:
Test Case Selection
Stratified random sampling across demographics, test types, and clinical conditions. Ensures validation covers edge cases, not just common scenarios.
Physician Ground Truth
Each case interpreted by 2+ board-certified physicians. Disagreements resolved by consensus with specialist consultation when needed.
Multi-Dimensional Scoring
Accuracy measured across: abnormality detection, clinical significance assessment, correlation identification, and recommendation appropriateness.
Continuous Monitoring
Production accuracy tracked via physician feedback loops. Model performance dashboards available to enterprise customers.
Include these questions in your vendor evaluation: (1) What is your validation methodology and sample size? (2) Who performed the validation (internal vs. third-party)? (3) How is accuracy defined and measured? (4) What is your process for continuous validation in production? (5) Can we review the full validation report?
ROI Model: Cost-Benefit Analysis Framework
Quantifying the return on investment for AI lab test interpretation software requires understanding both direct cost savings and indirect value creation. This framework helps build the business case for enterprise deployment.

📊 ROI Calculator Framework
Key metrics for enterprise AI lab interpretation deployment
Example: Mid-Size Hospital (500 beds, 150,000 lab tests/year)
| Cost/Benefit Category | Calculation | Annual Value |
|---|---|---|
| Physician Time Savings | 150K tests × 2 min saved × $3/min physician cost | $900,000 |
| Reduced Clarification Calls | 65% reduction × 30K calls/year × $15/call | $292,500 |
| Patient Satisfaction Impact | HCAHPS improvement → reimbursement bonus | $150,000 |
| Software License + Integration | Enterprise license + implementation | ($180,000) |
| Net Annual Benefit | $1,162,500 |
Value Creation Categories
Direct Time Savings
Physician interpretation time reduction is the most quantifiable benefit. Measure current interpretation time per result and project savings based on vendor-reported automation rates.
Reduced Support Burden
Patient-friendly reports reduce "what does this mean?" calls to nursing staff and physicians. Track call volume before/after implementation for concrete metrics.
Patient Satisfaction
Improved result communication correlates with HCAHPS scores, affecting value-based reimbursement. Difficult to quantify directly but strategically significant.
Competitive Differentiation
Patient experience differentiation in competitive markets. Particularly relevant for health systems competing for commercially insured patients.
RFP Procurement Checklist: 50 Essential Questions
This comprehensive checklist provides the essential questions to include in your Request for Proposal (RFP) when evaluating AI lab test interpretation software vendors. Use this as a scoring framework to objectively compare vendor responses.

📋 Enterprise RFP Checklist
50 critical evaluation criteria organized by category
🔗 Integration Capabilities
- HL7 v2.x message support (ORU, ORM, ADT)
- FHIR R4 native API implementation
- Epic App Orchard certification status
- Cerner CODE program participation
- Meditech Expanse integration support
- SMART on FHIR app capability
- CDA/C-CDA document generation
- RESTful API documentation quality
🔒 Security & Compliance
- HIPAA BAA availability
- SOC 2 Type II report (current year)
- GDPR DPA terms
- CE marking / MDR classification
- ISO 27001 certification
- Data residency options (US, EU, regional)
- Encryption standards (at rest, in transit)
- Penetration testing frequency/results
🎯 Clinical Accuracy
- Validation methodology documentation
- Accuracy metrics by test category
- Peer-reviewed publication references
- Third-party validation audits
- Continuous monitoring dashboards
- Physician feedback integration
- Model update validation process
- Edge case handling documentation
👨⚕️ Clinical Workflow
- Physician review/approval workflow
- Clinician override capability
- Critical value alerting integration
- Audit trail completeness
- Historical trending capability
- Multi-language report support
- Patient portal integration
- White-label customization depth
🏢 Enterprise Requirements
- Multi-facility support
- Multi-tenant architecture
- Role-based access control (RBAC)
- Single sign-on (SSO) support
- SLA commitments (uptime, response)
- Disaster recovery capabilities
- Scalability demonstration
- Reference customer availability
💰 Commercial Terms
- Pricing model transparency
- Volume discount structure
- Implementation cost breakdown
- Training and support inclusions
- Contract term flexibility
- Exit clause and data portability
- Price protection guarantees
- Vendor financial stability
Get the full 50-question RFP template with scoring rubric in editable format. Contact Us for RFP Template →
Vendor Comparison Framework
This framework provides an objective structure for comparing AI lab reporting software vendors. We present categories rather than specific competitor names to focus on capability evaluation rather than marketing claims.

Capability Comparison Matrix
| Capability | Category A (At-Home Wellness) |
Category B (Upload Tools) |
Category C (Enterprise) |
Kantesti Enterprise |
|---|---|---|---|---|
| HL7/FHIR Integration | ✗ Not Available | ✗ Not Available | ✓ Core Feature | ✓ Full HL7 v2.x + FHIR R4 |
| Physician Review Workflow | ✗ N/A | ◐ Limited | ✓ Standard | ✓ Configurable Workflows |
| Biomarker Coverage | 50-200 | 500-2,000 | 5,000-15,000 | 15,000+ |
| White-Label Capability | ✗ No | ◐ Limited | ✓ Available | ✓ Full Customization |
| Multi-Language Reports | 1-5 | 5-20 | 20-50 | 75+ Languages |
| HIPAA Compliance | ◐ Varies | ◐ Varies | ✓ Required | ✓ BAA Available |
| SOC 2 Type II | ✗ Rare | ◐ Some | ✓ Expected | ✓ Current Report |
| Enterprise SLA | ✗ No | ✗ No | ✓ Standard | ✓ 99.9% Uptime |
| Typical Use Case | Individual Consumers | Small Practices | Health Systems | Labs, Hospitals, Insurers |
Before detailed vendor comparison, confirm you're evaluating vendors in the appropriate category. Comparing a Category A (consumer wellness) vendor against enterprise requirements will produce misleading results. Match vendor category to your deployment requirements first.
Why Kantesti Enterprise: Evidence-Based Evaluation
Kantesti's B2B Platform serves as a reference implementation throughout this guide. This section provides specific evidence supporting its enterprise capabilities, while acknowledging appropriate limitations and considerations.

Kantesti Enterprise Capabilities
2.78 Trillion Parameter AI Model
Purpose-built neural network trained on 100M+ anonymized lab cases. Achieves 98.7% accuracy validated against physician consensus across 100,000+ test cases.
Native HL7/FHIR Integration
Production-proven integrations with major LIS and EHR platforms. Full HL7 v2.x support (2.5.1, 2.7) plus FHIR R4 native API. SMART on FHIR app available.
75+ Language Support
Patient-facing reports generated in 75+ languages with medically-validated translations. Critical for diverse patient populations and international deployments.
Full White-Label Capability
Complete branding customization for patient-facing reports and portals. API-first architecture enables seamless embedding in existing digital health platforms.
Enterprise Security & Compliance
HIPAA compliant with BAA available. GDPR certified with EU data residency options. SOC 2 Type II attested. CE marked for EU medical device compliance.
Medical Advisory Board
50+ board-certified physicians across 12 specialties provide ongoing clinical oversight. All AI interpretations undergo physician validation before deployment.
Kantesti Case Study Metrics
📈 Enterprise Deployment Outcomes
Aggregated metrics from Kantesti Enterprise deployments
Kantesti provides clinical decision support and educational information—not medical diagnosis or treatment recommendations. All AI interpretations are designed to support, not replace, physician judgment. Healthcare organizations must maintain appropriate physician oversight workflows and clinical governance.
Ready to Evaluate Kantesti Enterprise?
Schedule a personalized demo with our enterprise team. We'll walk through integration requirements, provide a custom ROI analysis, and connect you with reference customers in your sector.
No commitment required • Custom integration assessment • Reference customers available
Technical Glossary: Key Terms Defined
Understanding the terminology is essential for effective vendor evaluation and stakeholder communication. This glossary defines key terms used throughout this AI lab test interpretation software buyer's guide.
📚 Enterprise AI Lab Interpretation Glossary
LIS (Laboratory Information System)
Software managing laboratory workflow, sample tracking, and result reporting. Primary integration point for AI interpretation systems.
EHR/EMR
Electronic Health/Medical Record. Patient health information repository. EHR typically implies interoperability; EMR is facility-specific.
HL7 (Health Level Seven)
Healthcare data exchange standard. HL7 v2.x is message-based (common in LIS); HL7 FHIR is modern API-based standard.
FHIR (Fast Healthcare Interoperability Resources)
Modern healthcare API standard using RESTful architecture. FHIR R4 is current production version; R5 emerging.
CDS (Clinical Decision Support)
Systems providing clinicians with knowledge and patient-specific information to enhance decision-making. AI lab interpretation is a CDS category.
SaMD (Software as a Medical Device)
Software intended to be used for medical purposes without being part of a hardware device. Regulatory classification under FDA/MDR.
BAA (Business Associate Agreement)
HIPAA-required contract between covered entity and vendor handling PHI. Essential for US healthcare deployments.
Audit Trail
Chronological record of system activities. Required for regulatory compliance, quality assurance, and liability documentation.
White-Label
Vendor product rebrandable with customer's identity. Essential for patient-facing applications maintaining brand consistency.
API (Application Programming Interface)
Technical interface enabling system communication. RESTful APIs are standard for modern healthcare integrations.
SMART on FHIR
Standard for launching healthcare apps from EHR systems. Enables app marketplace models (Epic App Orchard, Cerner CODE).
Critical/Panic Value
Lab result requiring immediate clinical attention. AI systems must escalate these appropriately.
FAQ: Enterprise Buyer Questions Answered
Answers to the most common questions from healthcare IT decision-makers evaluating AI lab test interpretation software for enterprise deployment.
AI lab test interpretation software uses machine learning to generate clinical interpretations of laboratory results—not just reference range flags. Traditional LIS reporting marks values as "high" or "low" based on thresholds. AI interpretation analyzes patterns across multiple biomarkers, considers patient demographics, identifies correlations, and generates narrative explanations of clinical significance. This transforms raw data into actionable insights for physicians and patient-friendly explanations for healthcare consumers.
Enterprise AI lab interpretation platforms integrate via industry standards: HL7 v2.x messages for LIS communication and FHIR R4 APIs for EHR integration. For Epic, look for App Orchard certification and SMART on FHIR capability enabling MyChart integration. For Cerner/Oracle Health, verify CODE program participation. Integration typically involves: (1) Result feed from LIS to AI platform, (2) Interpretation generation, (3) Results return to EHR for physician review, (4) Patient delivery via portal. Kantesti supports all major integration patterns with production-proven Epic and Cerner deployments.
Minimum requirements: HIPAA compliance with Business Associate Agreement (BAA) for US deployments, SOC 2 Type II attestation demonstrating security controls, and GDPR certification for EU data. For medical device regulatory status, CE marking under MDR is required for EU deployment as SaMD. ISO 27001 certification provides additional security assurance. Kantesti maintains all these certifications. Request actual certification documents—not just marketing claims—during vendor evaluation.
Evaluate validation evidence using this hierarchy: (1) Peer-reviewed publications provide highest credibility; (2) Third-party audits by clinical experts are strong evidence; (3) Prospective clinical studies with outcome tracking are valuable; (4) Retrospective validation against historical cases is common but less rigorous; (5) Internal testing only is insufficient. Request detailed methodology documentation including sample size, case diversity, ground truth definition, and measurement criteria. Kantesti's 98.7% accuracy is based on 100,000+ physician-verified cases with published methodology.
Timeline varies by integration complexity: API-only integration (adding AI to existing workflows via REST API) typically takes 4-8 weeks. HL7 bidirectional integration with LIS requires 8-16 weeks including interface development and testing. Full EHR integration with physician workflows and patient portal may take 12-24 weeks depending on EHR vendor and customization requirements. Kantesti provides dedicated implementation teams with defined project milestones. Plan for pilot deployment before organization-wide rollout.
Enterprise platforms implement configurable physician review workflows. Options include: (1) Review all—physician approves every interpretation before patient delivery; (2) Exception-based—AI flags abnormal or complex cases for review, routine results auto-release; (3) Spot-check—random sampling for quality assurance. All systems should support physician override (modifying AI interpretation) with complete audit trails documenting all interventions. Critical values always escalate for immediate physician attention regardless of workflow configuration.
ROI sources include: (1) Physician time savings—typically 40% reduction in interpretation time, translating to significant labor cost savings; (2) Reduced support burden—65% fewer patient clarification calls when results include clear explanations; (3) Patient satisfaction—improved HCAHPS scores affecting value-based reimbursement; (4) Competitive differentiation—patient experience advantages in competitive markets. A 500-bed hospital processing 150,000 tests/year typically sees $1M+ annual net benefit after software costs. Kantesti provides custom ROI calculators during enterprise evaluation.
Coverage varies by vendor. Consumer-focused platforms typically support only common tests (CBC, metabolic panels). Enterprise platforms like Kantesti support 15,000+ biomarkers including specialty panels (autoimmune, endocrine, oncology markers). For rare conditions, AI systems should: (1) Recognize when confidence is lower and flag for physician review; (2) Provide relevant differential considerations without overstepping; (3) Reference appropriate clinical guidelines. Always verify biomarker coverage for your specific test menu during vendor evaluation.
Key questions for vendors: (1) Update frequency—how often is the model retrained? (2) Validation process—what testing occurs before updates deploy? (3) Customer notification—are customers informed of changes? (4) Rollback capability—can updates be reversed if issues arise? (5) Continuous monitoring—how is production accuracy tracked? Kantesti maintains a Medical Advisory Board of 50+ physicians providing ongoing oversight, with quarterly model reviews and continuous accuracy monitoring dashboards available to enterprise customers.
AI interpretation should apply demographic-specific reference ranges (age, sex, ethnicity, pregnancy status) rather than generic population averages. Pediatric and geriatric populations have different normal ranges. Pregnant patients require specialized interpretation. Verify that vendors: (1) Support demographic-adjusted ranges; (2) Handle special populations appropriately; (3) Flag cases requiring additional clinical context. Kantesti's model is trained on globally diverse populations with demographic-specific interpretation logic.
Enterprise platforms mitigate error risk through: (1) Physician review workflows—clinician oversight before patient delivery; (2) Confidence scoring—flagging lower-confidence interpretations for review; (3) Audit trails—documenting all AI outputs and physician interventions; (4) Feedback loops—capturing physician corrections for model improvement. Liability typically follows clinical decision-making: AI provides decision support, but treating physicians retain clinical responsibility. Review vendor contracts for liability allocation and professional liability insurance requirements.
Beyond technical capabilities, evaluate: (1) Financial stability—funding, revenue trajectory, burn rate; (2) Customer concentration—diverse customer base reduces risk; (3) Product roadmap—alignment with healthcare industry direction; (4) Reference customers—speak with similar organizations about their experience; (5) Exit provisions—data portability and transition support if relationship ends. Kantesti is backed by Microsoft Founders Hub, NVIDIA Inception Program, and Google Cloud partnerships, providing enterprise stability assurance.
Key Takeaways: Enterprise AI Lab Interpretation Software
Match Vendor Category to Your Requirements
Enterprise deployments require Category C vendors with HL7/FHIR integration, physician workflows, and enterprise compliance. Don't evaluate consumer platforms against enterprise requirements.
Integration Is the Primary Success Factor
68% of AI lab interpretation pilots fail due to integration challenges. Prioritize integration capability evaluation alongside accuracy claims.
Validate Accuracy Claims Rigorously
Request validation methodology, sample sizes, and ground truth definitions. Peer-reviewed publications and third-party audits provide highest credibility.
Compliance Documentation Is Non-Negotiable
Require actual certification documents (SOC 2 reports, BAA templates, CE certificates)—not just marketing claims about compliance.
Physician Oversight Workflows Are Essential
AI augments, not replaces, physician judgment. Verify configurable review workflows, override capabilities, and comprehensive audit trails.
ROI Extends Beyond Time Savings
Quantify patient satisfaction improvements, reduced support burden, and competitive differentiation alongside direct physician time savings.
📋 Enterprise Evaluation Quick Reference
Related Enterprise Resources
🧠 AI Blood Test Analyzer: How Kantesti Achieves 99.84% Accuracy
🔬 AI Blood Test Analysis: 98.7% Accuracy & Clinical Standards
📊 Customer Success Stories: Real-World Implementation Results
🔗 API Documentation: Integration Guide for Developers
⚔️ Kantesti vs GPT Models: Best AI Blood Test Interpretation 2025
📖 Blood Test Interpretation with AI: Complete Expert Guide
👨⚕️ Medical Advisory Board: Expert Physicians Behind AI Analysis
🥗 158 IQ AI: Blood Tests Into Nutrition & Supplement Plans
Start Your Enterprise Evaluation Today
Join leading healthcare organizations using Kantesti Enterprise for AI-powered lab interpretation. Our enterprise team will guide you through integration assessment, compliance verification, and custom ROI analysis.
Serving 2M+ users • 127+ countries • HIPAA/GDPR compliant • 98.7% accuracy
About This Enterprise Buyer's Guide
Julian Emirhan Bulut
CEO & Founder, Kantesti - PIYA AI
"Enterprise AI lab interpretation isn't just about accuracy—it's about seamless integration with existing clinical workflows. We built Kantesti Enterprise to be the platform healthcare IT teams actually want to deploy."
Julian Emirhan Bulut is the founder and CEO of PIYA AI and Kantesti, pioneering AI-driven healthcare solutions serving over 2 million users across 127+ countries. Under his leadership, Kantesti developed partnerships with Microsoft Founders Hub, NVIDIA Inception Program, and Google Cloud to deliver enterprise-grade AI lab interpretation at scale.
Medically Reviewed by Prof. Dr. Hans Weber, MD
Senior Medical Advisor - Laboratory Medicine
Professor Weber is the Director of the Institute for Laboratory Medicine at Munich University Medical Center, overseeing one of Europe's largest clinical laboratories. With an MD from University of Munich Medical School (1990), Habilitation in Laboratory Medicine from Technical University of Munich (1998), and Fellowship in Molecular Diagnostics from University Hospital Frankfurt, he brings unparalleled expertise to laboratory AI validation. As former President of the German Society for Clinical Chemistry (2018-2020) and an ISO 15189 Lead Assessor for Medical Laboratories, his 120+ publications in clinical chemistry and the European Award for Laboratory Excellence (2021) ensure our AI system meets the highest international standards. Learn more about our Medical Advisory Board →
Sources and References
This AI lab test interpretation software buyer's guide was developed using information from authoritative healthcare technology and regulatory sources.
- Kantesti Medical Validation & Clinical Standards - AI accuracy methodology and physician verification process
- Kantesti B2B Platform - Enterprise capabilities and integration documentation
- Kantesti API Documentation - Developer integration guide and API reference
- HL7 FHIR Standard - Healthcare interoperability specification
- FDA AI/ML Medical Devices Guidance - Regulatory framework for medical AI software
- HHS HIPAA Guidance - Healthcare privacy and security requirements
- GDPR Official Resources - European data protection requirements